ZirChrom Newsletter vol. #7
Vol.# 7- “ZirChrom®-MS - A "Deactivated" RPLC Zirconia Column for LC/MS” (PDF)
Welcome to the seventh issue of ZirChrom's electronic newsletter. This newsletter issues about every six weeks and addresses specific benefits of ZirChrom's family of ultra-stable zirconia-based HPLC phases and other practical topics of interest.
In this volume we introduce ZirChrom's newest reversed-phase column, ZirChrom®-MS, to be unveiled November 17-19 at the Eastern Analytical Symposium (Booth #220) in Somerset, NJ, USA.
In a previous edition of the electronic newsletter the ZirChrom®-EZ zirconia-based RPLC column was introduced, which has a metal chelator group that effectively "deactivates" the Lewis acid sites on zirconia. ZirChrom®-EZ provides the traditional unique selectivity of zirconia while facilitating the buffer selection process in the pH range of 1-10. Refer to e-newsletter #4 or the ZirChrom®-EZ page for more complete information on this exciting product.
Excellent user feedback on ZirChrom®-EZ has now led to the introduction of another "deactivated" zirconia product - ZirChrom®-MS. ZirChrom®-MS is a surface deactivated, reversed-phase zirconia column designed specifically for LC-MS applications, particularly those involving basic pharmaceutical compounds. The following unique features make ZirChrom®-MS an ideal choice for today's LC-MS method developer:
Compatible with volatile, near neutral pH mobile phase buffers including ammonium acetate and formate.
Enhanced retention for basic pharmaceutical compounds compared to bonded phase C18 silica under LC-MS compatible operating conditions.
Very different chromatographic selectivity for basic drugs compared to bonded phase C18 silica using LC-MS conditions.
Improved peak shape and efficiency for basic drugs compared to bonded phase C18 silica using LC-MS conditions.
The ability to analyze basic, acidic or neutral pharmaceutical compounds, or mixtures of all three, simultaneously.
Low column bleed characteristics due to covalent bonding chemistry.

Surface Deactivation Testing of ZirChrom®-MS
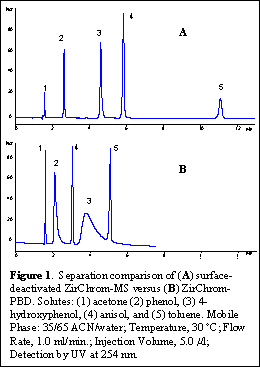
In order to demonstrate the new capabilities of ZirChrom®-MS, and compare it to some of our existing RPLC phases, we selected some small probe solutes that we typically use for quality assurance testing. Of particular note in the list of solutes is 4-hydroxyphenol, which is a strong Lewis base compound, and traditionally has required the use of a stronger harder Lewis base in the mobile phase to obtain good peak shape on zirconia-based columns. Figure 1 shows that excellent peak shape can be obtained for these small probe solutes on ZirChrom®-MS using no buffer in the mobile phase. The same separation is also shown in Figure 1, on a ZirChrom®-PBD column, which has not been exposed to any Lewis basic mobile phase additives (such as acetate, phosphate or fluoride containing buffers). Other common Lewis base moieties such as carboxylic acids, which can interact strongly with the non surface-deactivated zirconia columns, may be eluted on ZirChrom®-MS without the addition of nonvolatile phosphate containing buffers in the mobile phase.
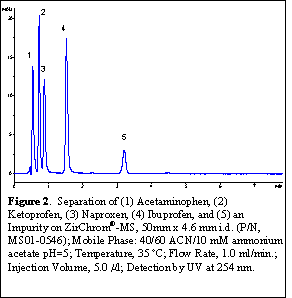
A good example where the Lewis acidity of zirconia-based supports for HPLC has historically presented problems is in the analysis of acidic pharmaceuticals such as non-steroidal anti-inflammatory drugs (NSAIDs), particularly in LC-MS applications where volatile mobile phase additives are required. While hard Lewis base type additives such as phosphate work well in applications with UV/Vis detection, their use is almost entirely prohibited in LC-MS applications due to their relatively low volatility. Here we demonstrate the utility of the new ZirChrom®-MS column for the analysis of this class of compounds using a very small amount (10 mM) of volatile buffer to assure good control of the mobile phase pH. Figure 2 shows the separation of a set of non-steroidal anti-inflammatory drugs, each containing carboxylate functionality, using simple acetonitrile/buffer isocratic elution and a LC-MS friendly acetate buffer. Excellent peak shape and efficiency is obtained for each component under these conditions.
Enhanced Retention for Basic Drugs of ZirChrom®-MS
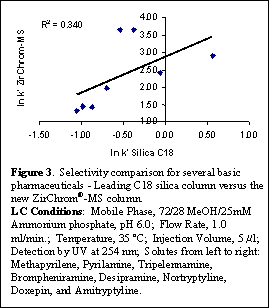
Another feature of the ZirChrom®-MS phase (as with the ZirChrom®-EZ phase) is its relatively higher retention for basic drugs in LC-MS compatible mobile phases compared to ZirChrom®-PBD. One typically obtains significantly higher retention for basic pharmaceutical compounds under the same conditions than on traditional bonded phase C18 silica owing to the mixed-mode of retention including both ion exchange and reversed-phase retention mechanisms. Figure 3 demonstrates this concept by comparing retention of some basic amine-containing pharmaceuticals on a ZirChrom®-MS column versus a leading C18 silica brand. Even though there is much higher carbon content to the silica-based column the ion-exchange contribution to retention results in higher retention factors on the ZirChrom®-MS column under identical LC conditions.
ZirChrom®-MS Exhibits Unique Selectivity for Basic
Pharmaceuticals
As a result of the mixed-mode ion exchange and reversed-phase characteristics of ZirChrom®-MS, the elution order of basic pharmaceuticals is often quite different compared to leading ODS phases. Figure 3 shows a plot of ln k' for eight common basic pharmaceuticals on a leading ODS phase versus ln k' for the same compounds on ZirChrom®-MS. There is no apparent correlation of the retention for these compounds on the ODS phase with the retention on ZirChrom®-MS. This different selectivity is particularly useful in method development for basic pharmaceuticals. When a pair of basic compounds cannot be separated using a traditional ODS phase, the chances of them separating on ZirChrom®-MS are much better than on another ODS phase.
ZirChrom®-MS Exhibits Excellent Efficiency and Peak Shape
for Basic Drugs Under Slightly Acidic LC-MS Compatible Mobile Phase Conditions
Amine-containing pharmaceuticals are well known for peak tailing on bonded phase silica C18 columns due to potential interactions between the amine functionalities and non-bonded residual silanol groups. The new ZirChrom®-MS column has excellent peak shapes for most basic compounds. For example, Figure 4 shows a separation of b-blockers on ZirChrom®-MS, which contain amine functionalities. The chromatogram shows excellent peaks shape and symmetry under LC-MS compatible conditions.
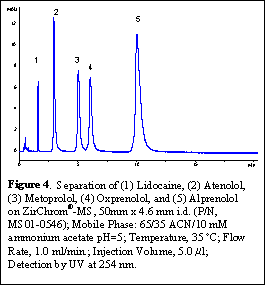
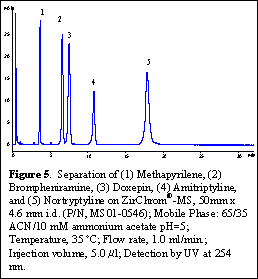
Figure 5 also shows a separation of a mixture of basic pharmaceuticals under LC-MS compatible mobile phase conditions.
Summary
ZirChrom®-MS represents another first of its kind zirconia-column designed specifically for MS detection and the high demands of pharmaceutical method development specifications. Using our novel covalently attached Lewis acid deactivation chemistry, ZirChrom has developed a highly retentive reversed-phase HPLC column, which is easy to use and which still has the inherent chemical stability advantages of zirconia-based HPLC columns. Most importantly this new column still maintains the very different chromatographic selectivity, especially for basic pharmaceuticals that zirconia-based columns are well known to have compared to traditional bonded C18 silica phases. This new column compliments the family of reversed phase columns that ZirChrom currently markets; a family of chemically different and thermally stable HPLC columns.
ZirChrom®-MS ordering and distributor information are available on the ZirChrom Separations website at: www.zirchrom.com. ZirChrom®-MS analytical and preparative scale HPLC columns are available in 3, 5, 10 and 25 micron particle sizes.
Also visit the ZirChrom website for more application notes using ultra-stable, high efficiency ZirChrom® columns, including the new ZirChrom®-MS.